|
|
 |
|
Calanoida ( Order ) |
|
|
|
Calanoidea ( Superfamily ) |
|
|
| |
| | | |
| Calanidae Dana, 1846 ( Calanoidea ) | | Ref.: | Giesbrecht, 1892 (p.41); Giesbrecht & Schmeil, 1898 (p.12); Sars, 1901 a (1903) (p.8); Esterly, 1905 (p.122); van Breemen, 1908 a (p.5, Genera Key); Wilson, 1932 a (p.19); Rose, 1933 a (p.55); Mori, 1937 (1964) (p.2, Genera Key); Brodsky, 1950 (1967) (p.81, 84); Vervoort, 1951 (p.55); Farran & Vervoort, 1951 (n°32, p.3); Chen & Zhang, 1965 (p.25); Björnberg, 1972 (p.24); Brosdsky, 1972 (1975) (p.1, 115, Rev.); Bradford & Jillett, 1974 (p.5, Rev.); Andronov, 1974 a (p.1005); Brodsky, 1976 (p.5); Vyshkvartzeva, 1976 (p.11); Björnberg & al., 1981 (p.616); Bowman & Abele, 1982 (p.10); Razouls, 1982 (p.57); Brodsky & al., 1983 (p.147); Sazhina, 1985 (p.107, 109, N); Fleminger, 1985 (p.273, 277, Table 2, Rem. A1); Mauchline, 1988 (p.719); Bradford, 1988 (p.73, Rev.); Zheng Zhong & al., 1984 (1989) (p.224, Genera Key); Nishida, 1989 (p.174, Rem.); Huys & Boxshall,1991 (p.461); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.22, Def.); Madhupratap & al., 1996 (p.863, Table 5: %/copepods); Chihara & Murano, 1997 (p.737); Barthélémy, 1999 a (p.30); Bradford-Grieve & al., 1999 (p.877, 902, 904, 905, 906, Genera Key); Ohtsuka & Huys, 2001 (p.445, 461); Boxshall & Halsey, 2004 (p.13; 49; 78: Def.; p.80: Genera Key); Mulyadi, 2004 (p.10, Genera Key for Indonesian Seas); Vives & Shmeleva, 2007 (p.889, Genera Key); Bradford-Grieve & Ahyong, 2010 (p.279, phylogeny, figs.4, 5); Blanco-Bercial & al., 2011 (p.103, Table 1, Fig.2, 3, 4, molecular biology, phylogeny); Laakmann & al., 2019 (p.330, fig. 2, 3, phylogenetic relationships); Hirai & al., 2020 (p.1, Fig.4: metabarcoding, Fig.8: OTUs distribution patterns, Fig.9: phylogenetic analysis)
Bradford-Grieve J.M., (2002 onwards). Key to calanoid copepod families. Version 1 : 2 oct 2002. http://www.crustacea.net/crustace/calanoida/index.htm  | | Rem.: | Fleminger (1985) studies the number and the arrangement of the setae and aesthetascs on the A1 of 25 species in the Calanidae family. The author defines 4 types of setae and 3 forms of aesthetasks. Besides the sexual dimorphism of the A1, which occur always in the (M) by a fusion of the 2 first proximal articles, and, according to the genera, the fusion of certain other articles, one observes in certain (F) of numerous species, an arrangement of groups of setae and aesthetascs homologous to those in the (M) in weak proportion and variable in time. These (F) having a (M) phenotype present two problems: one of systematic and phylogenetic interest, the other concerns the hypothesis of a sex change in the course of development and of its determinism. For the author the views of Brodsky (1972) as well as those expressed by Bradford et Jillett (1974) are not satisfactory.
Thereby Neocalanus is limited to 2 species N. gracilis and N. robustior likewise Calanus, sensu lato, holds the taxa of which the generic status is still uncertain (C. cristatus , C. hyperboreus , C. plumchrus , C. propinquus , C. simillimus , C. tonsus ). May be considered as Calanus sensu stricto the species grouped in the complex ' Calanus finmarchicus ' ( C. chilensis , C. finmarchicus , C. glacialis , C. pacificus C. orientalis , C. sinicus , C. australis , C. helgolandicus , C. marshallae ). The genus Nannocalanus is maintained, as well as those proposed by Bradford & Jillett (1974): Calanoides, Canthocalanus, Cosmocalanus, Mesocalanus, Undinula.
Bradford (1988) contests the scheme established by Brodsky (1972). She refers to the morphological relationships that permit the species groups more strict and more neutral concerning the phylogenetical relationships between the genera.
In addition to only the morphological criteria, various aspects of their biology and genome can now be contemplated (Bucklin, Frost & Kocher, 1992, 1995, Bucklin & Lajeunesse, 1994; Bucklin, Thomas & Kocher, 1996; Bucklin, Lajeunesse, Curry, Wallinga & Garrison, 1996).
Type-genus: Calanus Leach, 1816.
8 G.: Calanoides, Calanus, Canthocalanus, Cosmocalanus, Mesocalanus, Nannocalanus, Neocalanus, Undinula.
Key to genera after Boxshall & Halsey (2004, p.80) :
1 Coxa of P5 with row of small teeth along inner marin
2.
1 Coxa of P5 lacking ornamentation along inner margin
.. 4.
2 Male left P5 prehensile ; endopod rudimentary, lacking setae, exopod highly modified : basis of P2 and P°3 in female ornamented with spinules on posterior surface
. Cosmocalanus.
2 NeitherP5 prehensile in male ; basis of P2 and P3 in female lacking ornamentation of spinules on posterior surface
.. 3.
3 Male right P5 with inner margin setae on 3rd exopodal segment ; seminal receptacles fused in female
. Nannocalanus.
3 Male right P5 lacking inner setae on exopod ; seminal receptacles separate in female
Calanus.
4 2nd exopodal segment of P2 with deep invagination in outer margin ; male left P5 prehensile
Undinula.
4 Outer margin of 2nd exopodal segment of P2 not invaginated ; male left P5 not prehensile
. 5.
5 P1 with inner seta on basis modified into proximally expanded spine ; male mouthparts similar to those of female
Canthocalanus.
5 Inner basal seta on P1 plumose, not modified ; male mouthparts reduced
.. 6.
6 1st exopodal segment of P2 with recurved spinous process just medial to outer margin spine ; endopod of female P5 with 8 setae ; endopod of male right P5 with 8 setae
Neocalanus.
6 Spine on 1st exopodal segment of P2 not recurved ; endopod of female P5 with 6 or 7 setae ; endopod of male right P5 with less than 7 setae
.. 7.
7 Inner seta on 1st endopodal segment of P1 present ; endopod of female P5 with 6 setae ; endopod of male right P5 with 0 to 6 setae
. Calanoides.
7 Inner seta on 1st endopodal segment of P1 absent ; endopod of female P5 with 7 setae ; endopod of male right P5 with 7 setae
.. Mesocalanus. |  Issued from : A. Fleminger in Mar. Biol., 1985, 88. [p.277]. Morphological types of setae and aesthetascs. Nota: Four types of antennal setae may be recognized among the Calanidae: Type 1: smooth-surfaced, acuminate filaments varying from shorter than the bearing segment to more than 10 times its length (D), and located anteriorly on the segment. Type 2: short, coniform processes (E), always found on the anterior, distal end of Segment 8 and in many species on Segment 12. Type 3: pseudo-annulated setae bearing spike-like setules (F), located on Segments 22, 23 and 24. Type 4: short and with a more or less capitate apex, usually filled with lipid-like globules (G); they occur only in adults males on one or more of the following segments depending upon the species (fused Segment 1-2, Segment 7 and Segment 9. The capitate setae occupy the distal position that is filled by a Type 1 seta in the female and in the copepodite stage V. Aesthetascs:appear in 3 forms: Category 1: slender, vermiform processes (A) located on most segments of the female antenna. Category 2: digitiform, irregularly swollen or pinched along their length, and rounded at the apex (B); they occur on Segments 1 thriugh 9 in males. Category 3: occurring in males on Segments 10 to 23, vary structurally in fine detail among the genera, but all have a central cuticular keel articulating proximally with the antero-distal corner of the segment and extending to the succeeding antennal segment (C). A 'fleshy' layer covers the keel and extends as dorsal and ventral lamellae over the length of the Category 3 aesthetasc. These lamellae may be fused over the entire length of the aesthetasc or may separate at the distal end. The 'fleshy' layer is fragile and often is partially to totally eroded by capturing nets or during storage in formaldehyde. |
 Issued from : A. Fleminger in Mar. Biol., 1985, 88. [p.278, Table 2]. Calanidae Number of setae (Set) and aesthetascs (Aes) per antennule segment in the Calanidae genera examined. Individual segments with similar numbers of Set and Aes are grouped together. Arrangement of setae and aesthetascs: In all species studied, most or all of the adult females had a trithek arrangement (i.e. 1 proximal seta, 1 distal seta, and 1 distal aesthetasc) on each segment of A1 except Segments 1 and 20 to 25 (see Calanus pacificus californicus Fleminger, 1985). Segment 1 bears 3 setae and 1 aesthetasc. Segment 2, apparently a fusion of 3 segments, bears 3 such tritheks in positions 2a, 2b, and 2c. Segments 20 and 21 each lack 1 seta and the latter usually lacks the aesthetasc as well. Segments 22 and 23 have only 1 seta and 1 aesthetasc anteriorly and 1 posteriorly positioned, plumose, pseudoannulated seta (Type 3). Segment 24 has 2 setae similar to those of Segment 23, but no aesthetasc. Segment 25 bears 4 relatively short setae, i minute seta and 2 aesthetascs (see fig. A1 of calanus pacificus californicus and Calanoides philippinensis from Fleminger, 1985) ; it may be derived from the fusion of two or more segments. The A1 of adult males exhibits alternation of trithek and quadrithek combinations, the latter consisting of 2 setae and 2 aesthetascs and occurring on Segments 2b, 3, 5, 7 and 9. Segments 2a, 2c, 4, 6, 8 and 10 through 19 bear tritheks (as in the female).Segment 21 bears 1 seta and 1 aesthetasc in the male. The remaining segments carry the same numbers and types of setae and aesthetascs as found in the female. All the setae and th escale-like male aesthetascs are articulated and rotate through about 90° from normal to the antenna surface to flat against it. |
 Issued from : A. Fleminger in Mar. Biol., 1985, 88. [p.279, Table 2 (continued)]. Calanidae Number of setae (Set) and aesthetascs (Aes) per antennule segment in the Calanidae genera examined. Bracketed segments are completely fused; Segments 8 and 9 are incompletely fused. Individual segments with similar numbers of Set and Aes are grouped together. |
 Issued from : A. Fleminger in Mar. Biol., 1985, 88. [p.279, Table 3]. Calanus s.s. Antennule segmentation, setation and aesthetasc. Individual segments with similar numbers of Set and Aes are grouped together. Setae: Set = setiform; Spi = spiniform; Plu = plumose and pseudoannulated; Cap = capitate. Aesthetascs: Ver = vermiform; Dig = digitiform; Sca = scale-like. |
 Issued from : J.M. Bradford & J.B. Jillett in Crustaceana, 1974, 27 (1). [p.13, 14 fig.2]. Key to genera in Calanidae. 1 - Coxa of P5 inner edge with small teeth (fig.2i, j) ......... 2. 1 - Coxa of P5 inner edge naked (fig.2g, m) ......... 3. 2 - Basis of female P2 and P3 with posterior surface spines (fig.2c). Male left P5 prehensile (fig.2j) ........... Cosmocalanus. 2 - Basis of female P2 and P3 naked (fig.2d). Neither male P5 prehensile (fig.2i)....... Calanus. 3 - Exopod segment 2 of P2 in both sexes with outer proximal edge evaginate. Male left P5 prehensile (fig.2e,k) ............... Undinula. 3 - These characters absent ........... 4. 4 - Basis of P1 with distally directed seta on anterior surface modified into proximally thickened spine (fig.2a)................. Canthocalanus. 4 - Basis of P1 with distally directed setae of ordinary plumose type (fig.2b) ........... 5. 5 - Endopod of female P5 with 8 setae (fig.2f). Endopod of male right P5 with 8 setae (fig.2l) .............. Neocalanus. 5 - Endopod of female P5 with 7 setae (fig.2g). Endopod of male right P5 with 7 setae (fig.2m) ............. Mesocalanus. 5 - Endopod of female P5 with 6 setae (fig.2h). Endopod of male right P5 with no more than 6 setae (fig.2n) ......... Calanoides. |
 Issued from : G.A. Boxshall & S.H. Halsey in An Introduction to Copepod Diversity. The Ray Society, 2004, No 166, Part. I. [p.78]. Armature formula of swimming legs P1 to P4. Setation sometimes reduced. First exopodal segment of P2 with recurved spine in Neocalanus; 2nd exopodal segment of P2 with deep invagination in Undinula. Nota : Female P5 with 3-segmented rami as for legs P1 to P4, maximum setation as given in formula, often reduced. Inner margin of coxa P5 with row of teeth in some genera. - Male P5 weakly to strongly asymmetrical ; biramous with both rami typicalmly 3-segmented although in some genera one or both endopods are reduced or unsegmented. Right leg similar to P1 to P4 ; setation usually reduced by loss of inner margin setae from all exopodal segments. Setatio,n formula of endopod 0-1 ; 0-1 ; 2, 2, 2, often reduced. Left leg variable, often much longer than right leg. Exopod 3-segmented , as in P1-P4 in some genera, but lacking inner margin setae, to highly modified in Undinula and Cosmocalanus as complex grasping appendage. Eggs released into water. |
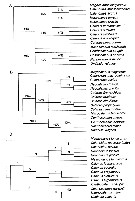 Issued from : J.M. Bradford-Grieve & S.T. Ahyong in J. Nat. Hist., 2010, 44 (5-6). [p.289, Fig.4]. (A) Strict consensus of six most parcimonious trees, length 81, consistency index = 0.58, retention index = 0.66, numbers above the branches indicate Jackknife support; (B) 50% majority-rule consensus of six most parcimonious trees; (C) single most-parcomonious tree derived from single round of successive weighting. Numbers above the branches are clade numbers. Megacalanus longicornis (Sars, 1905) (= Megacalanus princeps Wolfenden, 1904) is the outgroup. For the authors (p.294) the Calanidae probably evolved from a magacalanid-like ancestor. Although direct evidence of the ancestral habitat is lacking, the ancestral megacalanid was bathypelagic as are its modern relatives. It is suggested that the Calanidae radiated during the mid to late Triassic, possibly from the bathypelagic Megacalanidae (which may be adapted to low oxygen conditions) with high-latitude epipelagic locations being the first near-surface environments to be re-colonized. It is in surface environments that the metabolic rates of the Calanidae possibly adjusted to a well-oxygenated environment with a possible loss of their reliance on elevated of the level lactate deshydrogenase activity (LHD) (see Thuesen & al., 1998). Cladistic analysis of Calanidae recovered two major clades: a tropical epipelagic clade composed of species without ontogenetic vertical migration ( Canthocalanus + Cosmocalanus + Nannocalanus + Undinula); and a clade of ontogenetically migrating genera ( Neocalanus + Calanoides + Calanus + Mesocalanus) |
 Issued from : J.M. Bradford-Grieve & S.T. Ahyong in J. Nat. Hist., 2010, 44 (5-6). [p.292, Fig.5]. Clades derived from molecular analysis based on 18s ribosomal RNA. (A) Bucklin & al., 2003; (B) Taniguchi & al., 2004. | | | | | | (1) Calanoides Brady, 1883 | |
| | Syn.: | Carinocalanus Brodsky, 1972 (1975) (p.112) | | Ref.: | Brady, 1883 (p.74); 1914 (p.4); A. Scott, 1909 (p.10); Sewell, 1929 (p.25); Vervoort, 1946 (p.29); 1951 (p.55, Rem.); Carvalho, 1952 a (p.137); Tanaka, 1956 (p.259); Chen & Zhang,1965 (p.28); Ramirez, 1966 (p.6); Brodsky, 1972 (1975) (p.113, 114, 116); Bradford & Jillett, 1974 (p.6, 9, 13, Rev.); Fleminger, 1985 (p.273, 275, 278, 285, Table 2, Rem.: A1); Mauchline, 1988 (p.719); Razouls, 1982 (p.74); Bradford, 1988 (p.73,76, Rev.); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.23, Déf.); Chihara & Murano, 1997 (p.741); Mauchline, 1998 (p.68); Bradford-Grieve & al., 1999 (p.907: spp. Key); Boxshall & Halsey, 2004 (p.80) ; Vives & Shmeleva, 2007 (p.890); Sabatini & al., 2007 (p.341, Rem.: Comparison of species) | | Rem.: | type: Calanoides patagoniensis Brady,1883, not C. carinatus Kröyer,1849.
6 spp.
Redefinition from Bradford & Jillett (1974, p.9) :
- Cephalon and 1st thoracic segment partly fused.
- A1 male with segments 3 to 5 fused, though weak lines of separation are usually apparent.
- Mx2: 1st lobe with 5 setae.
- Mxp with fewer setae than in other calanids: terminal 5 segments with 4, 3, 2, 3 and 4 setae respectively.
- Male mouthparts reduced.
- Swimming legs without modification or ornamentation.
- 1st basipod (= coxopodite) of P5 with naked inner border in both sexes. Female P5 endopod with 6 setae. Male P5 endopods often reduced and unsegmented but never with more than 6 setae.
- Among the species of this genus, C. acutus has the least modified male mouthparts and C. philippinensis the most modified mouthparts. | | Remarks on dimensions and sex ratio: | | The mean female size is 3.218 mm (n = 10; SD = 1.1650), and in the male 3.008 mm (n = 9; SD = 0.8938). The size ratio male: femelle is 0.935. The sex ratio (F:M) is 1. | | | | | | Syn.: | Monoculus Gunnerus,1770; Cyclops (part.) Müller,1776;
Cetochilus Roussel de Vauzème,1834;
Nannocalanus (part.) Sars,1925 | | Ref.: | Kröyer, 1848 (in Damkaer & Damkaer, 1979, p.12); Brady, 1878 (p.37); Giesbrecht, 1892 (p.45, 88); Dahl, 1894 (p.74, spp. Key); Giesbrecht & Schmeil, 1898 (p.12, spp. Key); Wheeler, 1901 (p.164); Sars, 1901 a (1903) (p.8); Esterly, 1905 (p.123, clé spp.); van Breemen, 1908 a (p.6, spp. Key); A. Scott, 1909 (p.7); Wolfenden, 1911 (p.190); Esterly, 1924 (p.83); Sars, 1925 (p.5); Sewell, 1929 (p.19, Rem.); Wilson, 1932 a (p.19, spp. Key); Rose, 1933 a (p.55, spp. key); Mori, 1937 (1964) (p.11, spp. Key); Vervoort, 1946 (p.22); Brodsky, 1950 (1967) (p.85, clé spp.); Tanaka, 1956 (p.251); Brodsky, 1959 a (p.1548, spp. Key, Biogeography); 1961 (p.5, spp. Key); Chen & Zhang, 1965 (p.25); Ramirez, 1966 (p.5); Marshall & Orr, 1972 (p.155 & suiv.); Sheldon & al., 1972 (p.327, fig. 13: size particle vs. production's rate) Brodsky, 1972 (1975) (p.4, 112, 115, Rev., S/G & spp. Keys); Vyshkvartzeva, 1972 (1975) (p.186); 1976 (p.11); Frost, 1974 (p.77); Bradford & Jillett, 1974 (p.5, Rev.); Kabata, 1979 (p.20); Razouls, 1982 (p.62); Gardner & Szabo, 1982 (p.135); Brodsky & al., 1983 (p.149, spp. Key); Van der Spoel & Heyman, 1983 (p.147); Zheng Zhong & al., 1984 (1989) (p.225, spp. Key); Fleminger, 1985 (p.277, Table 2, 3, 4, Rem.: A1); Mauchline,1987 (p.719); Bradford, 1988 ( p.73, Rev.); De Decker & al., 1991 (p.27); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.29, Def.); Bucklin & al., 1995 (p.655); Chihara & Murano, 1997 (p.738); Mauchline, 1998 (p.67); Bradford-Grieve & al., 1999 (p.907: spp. Key); Boxshall & Halsey, 2004 (p.80); Vives & Shmeleva, 2007 (p.892, spp. Key) | | Rem.: | type: Monoculus finmarchicus Gunnerus, 1770. 14 spp.
Redefinition after Bradford-Grieve (1994, p.299) :
- Cephalon and 1st thoracic segment partly fused.
- A1 male with segments 3 to 5 fused in some cases.
- Mouthparts the same in both sexes;
- Mx2 with 5 or 6 setae on the 1st inner lobe.
- Swimming legs without modification or ornamentation.
- P5 with a toothed inner border on the 1st basipods (= coxopodite) in both sexes.
- Female P5 endopod with 8 setae;
- Male P5 with both rami 3-segmented, both endopods with 8 setae.
Marshall & Orr (1972, p.159-166 & litt.) underline the role of this genus in the food web, particularly as source of food for various planktonic forms and fishes, euphausids, whales, basking shark (Cetorhinus maximus), and even sea birds. In Antarctic, the chief food during the polar summer of Euphausia superba are copepods. In the Arctic other euphausids ( Thysanoesssa, Meganyctiphanes) or Calanus or a mixture. On the Norwegian coast Balaenopterus borealis (Sei whale) lives chiefly on copepods, especially Calanus finmarchicus (Hjort & Ruud, 1929; Hjort, 1933). For whales F.C Fraser (pers. comm.) has calculated thet the mouth of a right whale has a capacity of 1.5 m3, and that the corresponding figure for a rorqual is 2-3 times as much. The basking shark can filtered 2000 m3 per hour through the gill rakers. | | Remarks on dimensions and sex ratio: | | The mean female size is 3.847 mm (n = 28; SD = 1.6840), and in the male 3.568 mm (n = 28; SD = 1.1813). The size ratio (male: female) is 0.928. The sex ratio (F: M) is 1. | | | | | (3) Canthocalanus A. Scott, 1909 | |
| | Ref.: | A. Scott, 1909 (p.8); Vervoort, 1946 (p.36); Tanaka, 1956 (p.259); Chen & Zhang, 1965 (p.29); Brodsky, 1972 (1975) (p.114, 116, Rev.); Bradford, 1972 (p.14); Bradford & Jillett, 1974 (p.6, 10); Razouls, 1982 (p.76); Zheng Zhong & al., 1984 (1989) (p.227); Fleminger, 1985 (p.273, 278, Table 2, Rem.: A1); Mauchline, 1987 (p.719); Bradford, 1988 (p.73, 76, 79, Rem.); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.33, Déf.); Chihara & Murano, 1997 (p.739); Mauchline, 1998 (p.68); Boxshall & Halsey, 2004 (p.80) | | Rem.: | type: Calanus pauper Giesbrecht, 1888. 1 sp.
Redefinition after Bradford & Jillett (1974, p.10) :
- Cephalon and 1st thoracic segment fused.
- Mouthparts same in both sexes;
- Mx2 with 4 setae on the 1st inner lobe.
- P1: 1st basipod (= coxopodite) anterior margin terminates in a well defined projection; 2nd basiopod with distal seta on anterior surface modified into a proximally thickened spine.
- P2 to P4 without modification or ornamentation.
- P5 with the inner border of the 1st basipod (= coxopodite) naked in both sexes.
- Female P5 endopod with 7 setae. Male P5 with both rami 3-segmented, hardly modified on right; left endopod with only 2 terminal setae.
- For the authors, this genus occupies an intermediate position between those genera with a dentate inner border to the P5 coxopodite and also with similar mouthparts in both sexes, and those genera with a naked inner border to the P5 coxopodite and reduced male mouthparts. | | Remarks on dimensions and sex ratio: | | For one species only in the genus, concerning 21 specimens measured in the world: The mean female size is 1.526 mm (n = 30; SD = 0.1872), and the mean male size is 1.388 mm (n = 30; SD = 0.1843). The size ratio (male : female) is 0.90. The sex ratio (female : male) is possibly = 1 (17 females vs 19 males noted) | | | | | (4) Cosmocalanus Bradford & Jillett, 1974 | |
| | Ref.: | Bradford & Jillett, 1974 (p.6, 12); Razouls, 1982 (p.83); Fleminger, 1985 (p.273, 278, Table 2, Rem.: A1); Mauchline, 1987 (p.719); Bradford,1988 (p.74, 75, 79); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.35, Def.); Chihara & Murano, 1997 (p.737); Mauchline, 1998 (p.67: F, M); Barthélémy, 1999 a (p.26); Boxshall & Halsey, 2004 (p.80) | | Rem.: | type: Undina darwini Lubbock,1860. Total: 2 spp.
Definition from Bradford & Jillett (1974, p.12) :
- Cephalon and 1st thoracic segment fused.
- A1 of male with segments 3 to 5 fused.
- Postero-lateral corners of cephalothorax pointed in female.
- Mouthparts similar in both sexes.
- 1st inner lobe of Mx2 with 5 setae.
- Basal segments of the swimming legs ornamented with spines; these spines on the anterior surface of 1st basisipods of P1, and P3 to P5 in the famale; and P1, P3 and P4 in the male; these spines also on the posterior surface of 2nd basipods of P2 and P3.
- P5 of both sexes with teeth on the inner borders of the 1st basipods.
- Female P5 endopods with 7 setae.
- Male P5 with 1-segmented, rudimentary left endopod; left exopod highly modified compared with Calanus type: 1st exopodal segment normal; 2nd segment external spine very elongate with inner peg-like projection: 3rd segment elongate and complicated. Right leg not modified. | | Remarks on dimensions and sex ratio: | | The mean female size is 1.985 mm (n = 4; SD = 0.5533), and in male 1.750 mm (n = 4; SD = 0.5050). The size ratio (Male: Female) is 0.882. The sex ratio (Female : Male) is 1. | | | | | (5) Mesocalanus Bradford & Jillett, 1974 | |
| | Syn.: | Neocalanus (part.): Brodsky & al., 1983 (p.184, clé spp.) | | Ref.: | Bradford & Jillett, 1974 (p.6, 12); Razouls, 1982 (p.79); Gardner & Szabo, 1982 (p.147); Fleminger, 1985 (p.273, 278, Table 2, Rem.: A1); Bradford, 1988 (p.74-76); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.38, Def.); Chihara & Murano, 1997 (p.741); Mauchline, 1998 (p.68); Boxshall & Halsey, 2004 (p.80); Vives & Shmeleva, 2007 (p.897) | | Rem.: | For Brodsky (1980: pers. comm.) the creation of this genus appears not well guaranteed. Type : Calanus tenuicornis Dana,1849. Total: 2 spp.
Definition from Bradford & Jillett (1974, p.12) :
- Cephalon and 1st thoracic segment separate.
- A1 with segments 8 and 9 fused in female; in male segments 3 to 5, 7 and 8, and 9 and 10, fused.
- Male mouthparts reduced.
- Mx2 with 6 setae on the 1st inner lobe.
- Swimming legs P1 to P4 without modification or ornamentation.
- 1st basipod (coxopodite) of P5 with a smooth inner border in both sexes. P5 endopod of female with 7 setae. P5 male with both rami 3-segmented, endopods with 7 setae.
- For the authors, this genus is intermediate between Neocalanus and Calanoides. | | Remarks on dimensions and sex ratio: | | The mean female size is 2.520 mm (n = 4, SD = 0.8765), and the mean male size is 2.070 mm (n = 4, SD = 0.4976). The size ratio male: female is 0.821. The sex ratio female: male is 1. | | | | | (6) Nannocalanus Sars, 1925 | |
| | Syn.: | Canthocalanus (part.) : Brodsky, 1972 (1975) (p.116);
Calanus (part.) : Bradford & Jillett, 1974 (p.6, 9) | | Ref.: | Sars, 1925 (p.9); Vervoort, 1946 (p.25); Chen & Zhang, 1965 (p.30); Razouls, 1982 (p.79); Zheng Zhong & al., 1984 (1989) (p.227); Fleminger, 1985 (p.273, 278, Table 2, Rem.: A1); Bradford, 1988 (p.76, 79, Rem.); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.39, Def.); Chihara & Murano, 1997 (p.737); Barthélémy, 1999 a (p.26); Boxshall & Halsey, 2004 (p.80); Vives & Shmeleva, 2007 (p.899) | | Rem.: | type: Cetochilus minor Claus,1863. Total: 2 spp.
Definition after Bradford-Grieve (1994, p.39) :
- Cephalon and pedigerous segment 1 fused in both female and male. Otherwise very similar to Calanus.
- Right and left spermathecae fused on female genital segment.
- Male A1 with segments 1-2, 3-5, 24-25 fused.
- Male right P5 like other swimming legs, i.e, with setae on the inner border of the exopod, left endopod without setae, left exopod with outer edge spines greatly elongated.
- For the authors, it was incorrect in treating Nannocalanus as a synonym of Calanus (Bradford & Jillett, 1974, p.9) because N. minor would have been the only male in Calanus which had the exopod of the right P5 like the other swimming legs. That is, it has inner edge setae. Also the male P5 has a reduced number of setae or spines on the endopods and left exopod segment 3 compared with Calanus. The female of N. minor would have been the only female with the right and left spermathecae apparently fused (Brodsky, 1972). Brodsky (1972) considers Nannocalanus to be a junior synonym of Canthocalanus and erroneously assigns N. minor as its type species. This conclussion cannot be supported. | | Remarks on dimensions and sex ratio: | | The mean female size is 1.950 mm (n = 4; SD = 0.4564), and in male 1.633 mm (n = 4; SD = 0.3839). The size ratio male: female is 0.837. The sex ratio is 1. | | | | | (7) Neocalanus Sars, 1925 | |
| | Syn.: | Tropocalanus & Neocalanus (part.) Brodsky, 1972 (1975) (p.116); Brodsky & al., 1983 (p.184) | | Ref.: | Sars, 1925 (p.7); Sewell, 1929 (p.26, Rem.); Wilson, 1932 a (p.27); Vervoort, 1946 (p.38, Rem.); Oliveira, 1946 (p.454); Tanaka, 1956 (p.261); Chen & Zhang, 1965 (p.26); Bradford & Jillett, 1974 (p.10, Rev.); Razouls, 1982 (p.77); Gardner & Szabo, 1982 (p.141); Zheng Zhong & al., 1984 (1989) (p.227); Fleminger, 1985 (p.273, 278, Table 2, Rem.: A1); Mauchline, 1987 (p.719); Bradford, 1988 (p.74, 76, 79); Miller, 1988 (p.226, 270); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.41, Def.); Chihara & Murano, 1997 (p.739); Mauchline, 1998 (p.68); Bradford-Grieve & al., 1999 (p.908: spp. Key); Miller, 2002 (p.139); Boxshall & Halsey, 2004 (p.80); Vives & Shmeleva, 2007 (p.901, spp. Key); Ohtsuka & Nishida, 2019 (p.575, Fig.22.2, biogeography ) | | Rem.: | type: Calanus gracilis Dana,1849. Total: 6 spp.
Redefinition after Bradford & Jillett (1974, p.10) :
- Cephalon and 1st thoracic segment fused or separate.
- A1 male may have segments 24 and 25 fused.
- Male, and sometimes female, mouthparts reduced; Mx2 with 6 setae on 1st inner lobe.
- P2, in both sexes, with a recurved spine at the outer distal border of the 1st exopodal segment. 1st basipod (= coxopodite) of P5 with inner border naked in both sexes.
- Male P5 with both rami 3-segmented; left leg modified, endopod usually with 8 setae; right leg unmodified, or with inner edge setae of exopod absent.
- For the authors, N. gracilis and N. robustior are very closely related, both having the cephalon and 1st thoracic segment fused; the A1 male with segment 25 very small and fused to segment 24. N. plumchrus has the greatest mouthpart reduction in the both sexes.
After Ohtsuka & Nishida (2019, p.575), the divergence and colonization of Neocalanus are deduced in accord with Cenozoic events (< - 55 million years) (Machida & al., 2006; Ohtsuka & al., 2009). The taxa showing antitropical distributions are characterized by adaptationj to seasonal phytoplankton blpoom and by ontogenetic vertical migration, consistent with their origin in the Oligocene (- 34-23 million years). In addition, The existence of three Northern Hemisphere species suggests that speciation could have occurred in marginal seas after the Miocene (- 23-5.3 million years) | | Remarks on dimensions and sex ratio: | | The mean female size is 5.062 mm. (n = 12, SD = 2.299), and the mean male size is 4.416 mm (n = 12, SD = 2.066). The size ratio male: female is 0.872. The sex ratio (F:M) is 1. | | | | | (8) Undinula A. Scott, 1909 | |
| | Syn.: | Undina (part.) Dana,1847 | | Ref.: | A. Scott, 1909 (p.16); Sars, 1925 (p.10); Wilson, 1932 a (p.29); Dakin & Colefax, 1940 (p.87, clé spp.); Vervoort, 1946 (p.72); Carvalho, 1952 a (p.138); Tanaka, 1956 (p.264); Chen & Zhang, 1965 (p.30); Brodsky, 1972 (1975) (p.114, 116, 117, 120); Bradford & Jillett, 1974 (p.11, Redef.); Razouls, 1982 (p.80); Zheng Zhong & al., 1984 (1989) (p.228); Fleminger, 1985 (p.273, 278, Rem.: A1); Mauchline, 1988 (p.719); Bradford, 1988 (p.74, 77); Razouls, 1993 (p.308); Bradford-Grieve, 1994 (p.44); Chihara & Murano, 1997 (p.739); Mauchline, 1998 (p.68); Boxshall & Halsey, 2004 (p.80); Vives & Shmeleva, 2007 (p.905) | | Rem.: | Type: Undina vulgaris Dana,1849. Total: 1 sp.
Redefinition after Bradford & Jillett (1974, p.11) :
- cephalon and 1st thoracic segment fused.
Female thoracic corners extend postero-laterally into 1 or 2 points.
- Mouthparts similar in both sexes; Mx2 1st inner lobe with 6 setae.
- P2 with invaginate outer proximal border of the 2nd exopodal segment in both sexes.
- P5 1st basipod (= coxopodite) with inner border naked in both sexes; Female P5 endopod with 7 setae; those on the 1st and 2nd segments spine-like. Male left P5 hihhly modified; outer edge spines of 1st and 2nd exopodal segments very elongate and the 3rd segment very modified; endopod absent. Male right P5 with both rami 3-segmented, endopod with reduced setation, 2nd exopodal segment with outer distal border elongate extending as far as the 1st outer spuine of segment 3.
- The authors have removed U; darwini Lubbock,1860 and U. caroli Giesbrecht,1888 from this genus as they both differ from Undinula vulgaris as follows: the inner borders of the basipod of P5 are toothed; P5 male though superficially similar, are modified in a completely different manner; the outer margins of the 2nd exopods of P2 are not invaginate; and the setae on the two basal endopodal segments of the female P5 are not thickened and spine-like. | | Remarks on dimensions and sex ratio: | | The mean female size is 2.52 mm (n = 2; SD = 1.025), and for male 2.610 mm (n = 2; SD = 0.8697). The ratio size (male: female) is 1.036. The sex ratio (Female : Male) is 1. | | | | | | | Remarks on sizes :
The minimum and maximum values of the adult females of Calanidae family sare presented in figure 1 (in ordinates: total length in mm; in abscissa : Calanoides from 1 to 5, Calanus from 6 to 19, Canthocalanus : 20, Cosmocalanus : 21 and 22, Mesocalanus : 23 and 24, Nannocalanus : 25; Neocalanus from 26 to 31; Undinula : 32
Figure 1 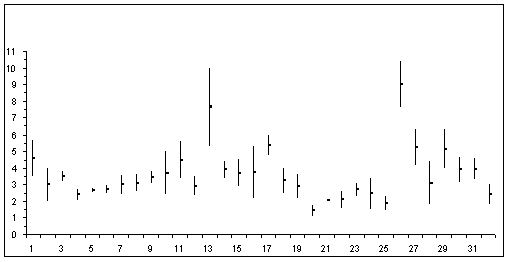
Table I: species, length, depth and location characteristics (E: epipelagic; M: meso-; B: bathy-).
species |
Lg (mm) |
|
|
Neocalanus cristatus |
7,50 à 10,50 |
Arct. & sub-arct. (Pacif.) |
E-B |
Calanus hyperboreus |
5,50 à 10 |
Arct. & sub-arct. (Atlant.) |
|
Calanoides acutus |
± 3,50 à ± 6 |
Antarct. & sub-antarct. |
E-M |
Calanus glacialis |
- |
Arct. & sub-arct. |
|
Calanus propinquus |
- |
Antarct. & sub-antarct. |
E-M |
Neocalanus flemingeri |
- |
sub-arct. (Pacif.) |
|
Neocalanus plumchrus |
- |
sub-arct. (Pacif.) |
|
Calanus pacificus |
2,15 à 5 |
sub-arct., cold temperate |
|
Neocalanus robustior |
- |
cosmop, tropic., subarct. |
E-M |
Neocalanus tonsus |
- |
sub-antarct., tropic. |
|
Calanus finmarchicus |
- |
cold temperate |
E-M |
Calanoides carinatus |
± 2,50 à ± 4 |
cosmop, sub-antarct, tropic. |
E-B |
Calanoides macrocarinatus |
- |
sub-antarct. |
E-B |
Calanus australis |
- |
sub-antarct. |
E-B |
Calanus chilensis |
- |
cold temperate |
|
Calanus euxinus |
- |
temperate |
|
Calanus helgolandicus |
- |
temperate |
E-M |
Calanus simillimus |
- |
sub-antarct. |
|
Calanus sinicus |
- |
tropic., cold temperate |
|
Calanus marshallae |
- |
sub-arct. |
|
Calanus jaschnovi |
- |
temperate |
|
Neocalanus gracilis |
- |
cosmop., topic., cold temperate |
|
Calanoides patagoniensis |
± 2 à ± 3 |
sub-antarct. |
E-?M |
Calanoides philippinensis |
- |
tropic. |
M |
Calanus agulhensis |
- |
temperate |
|
Cosmocalanus caroli |
- |
tropic. |
|
Cosmocalanus darwini |
- |
cosmop., topic., cold temperate |
E-?M |
Mesocalanus lighti |
- |
sub-tropic. |
|
Mesocalanus tenuicornis |
- |
cosmop., topic., temperate |
E-B |
Undinula vulgaris |
- |
cosmop., tropic. |
|
Nannocalanus minor |
- |
cosmop., tropic., cold temperate |
E-M |
Canthocalanus pauper |
- |
tropic. (Indo-Pacif.) |
E |
| | | | | | | | | | | |
|
|
 Any use of this site for a publication will be mentioned with the following reference : Any use of this site for a publication will be mentioned with the following reference :
Razouls C., Desreumaux N., Kouwenberg J. and de Bovée F., 2005-2026. - Biodiversity of Marine Planktonic Copepods (morphology, geographical distribution and biological data). Sorbonne University, CNRS. Available at http://copepodes.obs-banyuls.fr/en [Accessed March 15, 2026] © copyright 2005-2026 Sorbonne University, CNRS
|
|
 |
 |