Abstract | Introduction | Presentation | Diversity | Main marine currents | The different oceanic zones : Summary of the species - Marine currents and other maps | Cosmopolitanism and endemism | Species indicative of continental drift Species whose localization is difficult to explain | Anthropic mechanisms | Conclusion
1) Surface currents (
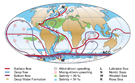
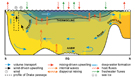
By definition, planktonic organisms cannot free themselves of ocean currents. Very early on the authors suggested that there was probably a correlation between the main surface- (best-known at the time) or deep-water currents and the geographical distribution of certain genera and species. Sewell (1948, p.387) classified copepods into two groups: epipelagic subjected to surface currents and meso- and bathypelagic subjected to intermediate and deep currents (Sewell, 1948, maps 1 & 2). Knowledge of bathymetric distribution is often imprecise because of sampling techniques and further complicated by circadian or ontogenic vertical migration and upwellings, which makes it very difficult to establish causality. This problem may be approached using a subset of the matrix associating the presence of species in the various zones with the route followed by certain currents (fig. C2). Most of the fairly cosmopolitan species probably fall under this schema. The geographical data covers little over a century but is the product of several millennia, or rather longer if we include the aspects of historical geology mentioned earlier The Agulhas Current (Boden & Parker, 1986; Hutchings et al., 1995) constitutes an essential exchange route between the Indo-Pacific and the Atlantic. The only barrier is a thermal one relative to surface-dwelling tropical species (Van der Spoel & Heyman, 1983, figs. 18, 19, 20) e.g. for Pontellina morii (Fleminger & Hulsemann, 1974, fig. 12) and an equatorial form such as Copilia lata. If we impose as the selection criteria a species' presence in zones 16, 5, 7, 8 and absence from the Arctic zone, ignoring all other zones, 145 meet these criteria. Sander & Moore (1979) explain the localisation of Dioithona oculata by this mode of transport, and this must also be the case for most of the species included above. Jones (1966 b), however, demonstrates that the southern tip of South Africa isolates the Indo-Pacific and Atlantic tropical populations of Candacia pachydactyla. The Atlantic and Pacific forms reveal morphological differences reflecting an imbalance in the genetic pool. Fleminger & Hulsemann (1987) demonstrate similar genetic gradients for populations of Calanus helgolandicus from the Atlantic to the Mediterranean and through to the Black Sea, as do Bucklin et al. (1996) for Calanus finmarchicus in the North Atlantic. The south equatorial Atlantic current splits near equatorial Brazil into one branch heading north towards the West Indies (zone 7) and another flowing south off the south coast of Brazil (Le Pichon et al., 1978; Angel, 1979; Boltovskoy, 1981). 119 species among the 145 in the preceding list are also found in this zone (zone 13). Most of the species carried by the Gulf Stream are found in the Sargasso Sea and the temperate zones of the eastern Atlantic (zone 8). The North-Atlantic Drift and the Irminger Current carry some of these species as far as southern Iceland and Greenland (Aksnes & Blindheim, 1996). The warm Kuroshio Current which flows along the Pacific part of the Philippines, the edge of the China Seas and the southern and central fringe of Japan continues towards the eastern Pacific and the Californian coast (North Pacific Drift). The percentage of species common to zones 22 and 25 reflects the faunal community's relationship to this means of transport. 333 species meet these conditions. Eucalanus californicus and Pareucalanus parki illustrate this distribution mode (Fleminger & Hulsemann, 1973) as do Pontellopsis yamadae and Mimocalanus heronae. The current generated by the northern subtropical anticyclonic gyre (North Equatorial Current) maintains this community of species (Beklemishev et al., 1972; Voronina, 1978; van der Spoel & Pierrot-Bults, 1979; Beklemishev, 1981). 41 species can enter the Gulf of Alaska sensu lato in the Alaska Current including: Acartia liljborgi, Aetideus bradyi, Euchirella bitumida, messinensis, pulchra, Gaetanus armiger, minor, pungens, Pachyptilus abbreviatus, Cosmocalanus darwini, Clausocalanus mastigophorus, Paraeuchaeta hanseni, tonsa, Lucicutia magna, Megacalanus princeps, Gaussia princeps, Metridia princeps, Acrocalanus gibber, gracilis, Archescolecithrix auropecten, Lophothrix frontalis, Scaphocalanus echinatus, Scolecithricella abyssalis, S. dentata, Scolecithrix danae, Scottocalanus helenae, infrequens, Spinocalanus brevicaudatus, Oithona setigera, Corycaeus affinis, C. catus, C. erythraeus, Conaea rapax, Oncaea media, Monothula subtilis, Sapphirina intestinata, Oculosetella gracilis.
2) Deep-water Currents (
Intermediate deep-water currents (Sewell, 1948, chart 2; van der Spoel & Heyman, 1983, fig. 24) give an insight into the localisation of certain species and in particular the bipolar forms sensu lato. The following Arctic and sub-Arctic forms are observed in temperate and tropical parts of the Atlantic, or in the Pacific: Chiridius obtusifrons (in zones 27-10-24-9-11), Xanthocalanus profundus (27-8), propinquus (27-9-11-6-16-5), Euaugaptilus hyperboreus (27-22-8-17), Spinocalanus polaris (27-23-9-7), Mimocalanus crassus (27-7-8-6-16), Paraeuchaeta norvegica (27-10-9-11-7-8-14-16), Bradyidius similis (27-10-23-24-9-11-8), Centropages abdominalis (27-23-24-22-25-21-14-19-26), Homeognathia brevis (27-9-8-14-17-26), Epicalymma schmitti (27-24-9-8-4), umbonata (27-24-9-8-4), Oncaea lacinia (27-23-24-9-14-4). Conversely, Antarctic and sub-Antarctic forms may be observed further North: Aetideopsis minor (4-3-13-27), Aetideus arcuatus (3-26-17-7-8-24), australis (4-3-26-12-18-25), Chiridiella subaequalis (4-8-23), Euchirella latirostris (3-12-18), rostromagna (4-3-26-13-18), similis (3-26-5-12-18-8), Gaetanus antarcticus (4-3-26-13-12-16-19-8-22-24), Pseudochirella hirsuta (4-3-26-12-16), mawsoni (4-3-26-18-16), spinosa (3-12-8), Augaptilus cornutus (4-3-16-8-11-23-24), Euaugaptilus aliquantus (4-3-13-16), maxillaris (4-3-16-6-17-8-22), perasetosus (4-3-26-13), placitus (4-26-17-8-22), Haloptilus ocellatus (4-3-13), Pontoptilus ovalis (4-16-6-8), Pseudaugaptilus longiremis (4-16-7-8-14-10), Calanus australis (3-26-13-18-16-20), Candacia cheirura (3-26-5-13-12-18-16-20), maxima (4-3-13-16), Farrania frigida (4-26-12-16-17-21-7-22), Rhincalanus gigas (4-3-26-13-18-16-20), Subeucalanus longiceps (4-3-26-5-13-18-16-6), Paraeuchaeta abbreviata (4-3-18-20-9), aequatorialis (4-3-26-12-16-20-19-17-21-8-22), antarctica (4-3-26-13-18-16), biloba (4-3-26-13-18-16-21), calva (3-12-16-17-21-7-22-11), comosa (3-26-13-12-18-16-19-17-21-7-22), confusa (3-26-21-7-22), dactylifera (4-3-13-12-18-19), exigua (4-3-12-18-16), kurilensis (4-3-12-18-16-20-19-22-23-9), malayensis (3-26-16-20-17-21-7-22), parvula (4-3-12-16), rasa (4-3-26-13-12-16), regalis (4-3-26-12-20-19), tumidula (4-3-20-17-21-7-8-22-23), Heterorhabdus austrinus (4-3-26-5-13-18-16-8), Lucicutia bradyana (3-26-12-18-16-19), Bathycalanus bradyi (4-3-26-5-12-16-6-19-17-25-22-23-24-9), Bradycalanus gigas (4-16), sarsi (3-26-16-8-11-10), typicus (4-3-26-5-16-20-19-17-7-8-25-22-9), Cephalophanes frigidus (4-3-26-5-13-18-16-20-6-19-17-15-21-7-8-14-25-22-11-23-9-10-27), Cornucalanus robustus (4-3-26-6), simplex (4-26-16-6-17-8), Onchocalanus hirtipes (4-7-8-22-9-10), magnus (4-3-26-16-7-22-23-24), Pseudoamallothrix obtusifrons (4-3-26-16-20-19-17-7-8-11), Amallothrix dentipes (4-3-26-13-12-18-16), A. robusta (4-3-26-20-6-17-8-9-10), Landrumius gigas (3-26-19-17), Mixtocalanus alter (4-3-21-8-22), Racovitzanus antarcticus (4-3-26-5-13-18-16-8-25-22-23-24-9-27), Scaphocalanus impar (4-13), S. major (4-3-18-16-6-17-21-7-8-22-23-24-9), Spinocalanus horridus (4-3-5-12-6-21-7-8-22-23-24-10-27), S. terranovae (4-3-5-14), Teneriforma naso (4-5-18-16-19-7-8), Homeognathia flemingeri (4-3-16-24), Atrophia minuta (3-26-21-7-8-14-24-9), wilsonae (4-24), Oncaea curvata (4-3-26-5-13). Sewell (1948, p.524, 528 & foll.) emphasised the importance of the Intermediate Antarctic Current in bringing species as far as the Gulf of Oman, for example Paraeuchaeta hanseni, Valdiviella insignis and Bathycalanus bradyi. The Intermediate Mediterranean Current from which stems the Lusitanian Current (Fraser, 1961 c; R. G. Johnson, 1997) allows Undinella stirni, described as Mediterranean where it is cited several times (Grice, 1971 b; Scotto di carlo et al., 1984; 1991), to be observed in the Sargasso Sea (Deevey & Brooks, 1977). This species is mesopelagic (1000 m et plus). Similarly Labidocera brunescens noted by Sars (1925) North of the Azores may also be an indicator. However, the Mediterranean origins of these species cannot be claimed with certainty.
|
|
Razouls C., Desreumaux N., Kouwenberg J. and de Bovée F., 2005-2026. - Biodiversity of Marine Planktonic Copepods (morphology, geographical distribution and biological data). Sorbonne University, CNRS. Available at http://copepodes.obs-banyuls.fr/en [Accessed March 22, 2026] © copyright 2005-2026 Sorbonne University, CNRS |